Although the very first balloons were hot air balloons, the difficulties in producing a reliable, controllable and safe supply of hot air were so great that hydrogen, and later helium, balloons very soon replaced them. The first Manned balloon flight in a hot air balloon was in November 1783, but within a few years hot air ballooning was dead, until the early 1960s.
The history of ballooning is discussed on another Page of this Web Site - to link to it please click here ![]()
The invention in 1960 of the light-weight propane burner introduced the reliable, controllable and safe supply of hot air which had been lacking in the 1780s and 1790s, and hot air ballooning came back from the dead. Today almost all recreational ballooning (ballooning for fun) is in hot air balloons. The light-weight propane burner is described at the end of this Web Page.
The density of air at sea level and 20oC is about 1.25 kg/m3, but at 80oC it is only about 1.05 kg/m3, a difference of 0.2 kg/m3. This means that at sea level a hot air balloon containing 1000 m3 of air at 80oC can lift about 200 kg, only about one fifth of what a helium balloon of the same size could lift.
You can buy model hot air balloon kits, or you can make your own small hot air balloon out of many different materials. The plastic cover your dry-cleaning comes back in is almost ideal because it will lift small loads when filled with warm air from a hair drier. A black dustbin liner is not suitable because it is made of such thick and heavy plastic that the temperature of the air needed to give it enough buoyancy is very close to the temperature at which it catches fire. Remember there are very strict laws about flying a hot air balloon with a burner attached to it.
A hot air balloon, like a helium balloon, moves at the speed of the wind in the direction of the wind. This is what makes ballooning so fascinating: there is no sensation of speed and no wind noise at all. The only control you have is over the the height (altitude) of the balloon. To gain height you turn the burner on. This heats the air in the envelope and increases its buoyancy - this gives the balloon an upward acceleration. Once the burner is turned off the air inside it will start to cool and the buoyancy will start to decrease. The upward acceleration will get less and then the balloon will start to accelerate downwards until the burner is turned on again. Unlike a helium balloon it is almost impossible to keep a hot air balloon at a constant height. Most hot air balloon flights therefore consist of a slow loss in height with the burner off followed by a much quicker gain in height with the burner on.
Landing a hot air balloon involves first allowing the air to cool enough to produce a downward acceleration, and then turning on the burner to reduce this acceleration so that the moment it hits the ground the height, downward velocity and downward acceleration are all zero at exactly the same time - but the burner must be off at the moment of impact, otherwise as the balloon tips over the envelope will catch fire! As with a helium balloon this is Not Easy!
A hot air balloon is open to the atmosphere at the bottom so the pressure inside it is only very slightly above atmospheric pressure, so unlike a helium balloon the envelope does not stretch as the balloon goes higher and atmospheric pressure gets less.
Propane is widely used as a fuel. It is usually stored as a liquid under pressure in a cylinder or tank. At atmospheric pressure and temperature the pressure in a propane tank is about 5 bars (five times atmospheric pressure). The storage of propane is discussed at greater length on the Page on Storage of Gases - to link to it click here ![]()
When we open the valve on the tank some gas will come out. Some of the liquid in the tank evaporates to take its place, ensuring a steady flow of gas out of the tank. However, we need heat to evaporate a liquid - you can prove this by moistening the back of your hand and then blowing on it. The water evaporates and cools your skin; this is why sweating cools you down. The heat needed to evaporate the propane usually comes from the liquid propane in the tank and its temperature drops. As the temperature goes down the rate at which the propane is evaporating will go down too.
If we are using propane for cooking in a caravan, or even supplying central heating to a house without a mains gas supply, we do not need huge amounts of heat, so the rate at which the gas evaporates, which is the rate at which we are using gas, is quite slow and the temperature drops only a little.
But if we need huge amounts of heat in a hurry, for example to warm an aircraft hangar or fill a hot air balloon, the amount of heat required to evaporate the gas needed is so great that if this heat came only from the liquid in the tank the temperature would fall so much that evaporation would stop. So to provide the heat needed to evaporate the liquid we use the heat provided by the burning gas!
We use a special syphon tank. This has an outlet at the top to provide a small and steady flow of gas to the pilot light. But it also has a pipe from the bottom carrying liquid propane under pressure. The liquid propane passes through a coil around the main burner flame, and the heat from the flame turns the liquid into a gas at a very high pressure. This high pressure gas is then burned in the main burner.
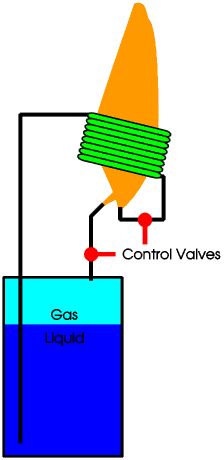

The Tilley® lamp and Primus® stove work on a similar principle, except that they burn paraffin, which is a liquid at room temperature. The paraffin is stored in a tank under pressure, the pressure being produced by pumping air into the tank. The liquid paraffin is passed through a coil round the flame and this vapourises it. The paraffin vapour then burns at a jet with a very hot blue flame. This produces far more heat and a far higher temperature than could be produced by burning the paraffin at a wick. In a Tilley lamp this blue flame then heats a mantle which produces a very bright white light.
Although Primus stoves and Tilley lamps are much more powerful and more economical that stoves or lamps burning paraffin at a wick they are also very much more expensive and complicated. Today most people needing powerful heating or lighting use a propane or butane burner rather than a Tilley lamp or Primus stove burning paraffin.
© Barry Gray August 2006